 This clues us into the structure of a molecule if there are multiple options: the one with the least/lowest formal charges is the preferred structure. Ideally, an atom in a molecule wants to have a formal charge of zero: this is the lowest energy, and thus the most stable state for it to be in. We will explore some examples of nonzero charge below. 
Note: though the formal charge in these two examples has been zero, that will not always be the case. But more on that later! How to Calculate Formal Charge:įormal Charge (FC) = (# of valence electrons) – (½)(number of bonded electrons) – (number of unbonded electrons) Examples:ĬH 3O: what is the formal charge on the carbon?Ĭarbon has 4 valence electrons, 8 bonded electrons (two single bonds and one double bond), and no unbonded electrons. This is a hypothetical measure, not a real representation of the actual charge on an atom, which looks at the ways electrons are actually shared between atoms in a bond. Resonance: a term used to describe the hybrid bonding in a molecule with multiple possible structures.įormal Charge is a charge assigned to an atom under the assumption that all electrons in bonds are shared equally.Molecule: a group of atoms bonded together.Ion: an atom or molecule with a net ionic charge, due to the presence or lack of electrons.Quantifying protons, neutrons, and electrons.In this tutorial, you will learn what is formal charge, how to calculate it, and its significance in practice. Clipart and elements found in this PDF are copyrighted and cannot be extracted and used outside of this file without permission or license.įiles are partially or fully non-editable to protect the images that are copyrighted and purchased through licenses.Formal charge is an essential, basic concept to master in order to better understand molecular structures and reactions. If you and a team of teachers would like to use this resource together, please purchase additional licenses on the resource purchase page.įailure to comply with these terms of use is a copyright infringement and a violation of the Digital Millennium Copyright Act (DMCA). You are not allowed to distribute this digital resource to other teachers or post this resource on any webpage or server that is available for public view. This resource is for you, the purchaser, alone. 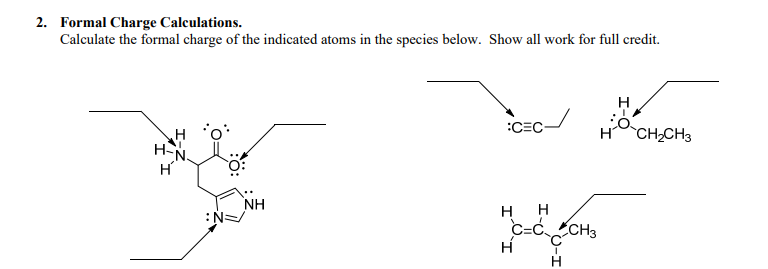
* place this file on your own password-protected class page or server (Blackboard, Google Drive, etc) AS LONG AS no other teacher has access to that class webpage. Purchasing my teaching resources allows you to: If you have any questions or concerns, please reach out to us on the question and answer section of my store and we will get back to you quickly! 
The PDF file contains the student page, the answer key, and my terms of use page.Ĭheck out my Chemistry Doodle Notes for some engaging resources that thoroughly explain Chemistry concepts in a versatile way! 
This page is also part of a Chemical Bonding Chemistry Homework Unit. Click here to read about that whole year bundle if you want to buy a whole year’s worth of homework pages at a discount as compared to buying them separately. This page is part of my giant Chemistry Homework for a Year Bundle. This page would be good for honors level learners. I recommend students use this page before they complete the more complex Lewis structure pages. This skill helps students to figure out the correct or "most correct" Lewis structure for a particular compound. This chemistry homework page is perfect for students to practice calculating the formal charge for atoms within a Lewis structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
